Abstract
Late spring and summer represent a critical ecological period for members of the Gray Treefrog and Cope’s Gray Treefrog species complex. During this time several energy goals must be met to ensure reproduction takes place followed by a period of preparation for winter dormancy. We hypothesized that the effects of this period could be measured by changes in weight, where weight in both sexes would decline during the reproductive part of the season (late spring/early summer), followed by an increase in weight for both sexes during the late summer in preparation for winter. We tracked a total of 97 male and 63 female treefrogs from four ponds and two lake inlets over a period of seven summers (2015–2021). We regressed weights of both sexes against ordinal dates for the summer reproductive period (May–June, 123–182) and winter preparatory period (July–August, 183–261). For both sexes we found a significant overall weight loss in the summer reproductive period (males: F1,129, P = 0.0013, females: F1,220, P = 0.0038) followed by a weight gain in the winter preparatory period (males: F1,317, P < 0.0001, females: F1,209, P < 0.0001) over the seven-year period. However, on a yearly basis, these significant weight change patterns were only seen in some years (summer reproductive, males: 42%, females: 17%; winter preparatory, males: 42%, females: 42%). We conclude a general pattern of weight loss during the reproductive period of the season followed by a general pattern of weight gain, but that this effect may be masked in some years by other ecological factors.
Introduction
Late spring and summer represent an important period for members of the Gray Treefrog and Cope’s Gray Treefrog species complex (Hyla versicolor, LeConte/chrysoscelis, Cope; hereafter treefrog). Most breeding takes place during late spring through mid-summer, and this energy-expensive period is followed by preparations for overwinter dormancy, another energetically expensive time (Smith et al., 2003; Briggler and Johnson, 2021). As such, the summer energy budget is an important component of the annual fitness of individuals in treefrog populations. This study explores the importance of this energy budget in terms of weight change in relation to both sexes over the breeding and winter preparatory time periods.
In northwest Missouri, treefrog breeding typically reaches its peak in early May and lasts through the end of June or early July, with small amounts of breeding extending even to August (Briggler and Johnson, 2021). Males will establish choruses at shallow ponds, or similar sites where fish are unlikely to prey on eggs, allowing females to approach and select mates based on site and call characteristics (Fellers, 1979; Schwartz et al., 2001). This is particularly energetically expensive for males, who may produce over 5000 calls a night (Wells and Taigen, 1986). However, females incur energy costs as well, traveling nightly to visit choruses and using fat supplies to produce eggs (Johnson et al., 2007; Rastogi et al., 2010). It should be no surprise then, that both males and females lose weight during this time (Rastogi et al., 2010).
During winter, dormancy typically occurs in leaf litter above the frost-line, where temperatures are moderated, but still often below freezing (Layne, 1999; Johnson et al., 2008). Cryoprotectants are secreted by the liver and take the form of glucose (Hyla chrysoscelis) broken down from glycogen (Hyla versicolor, Costanzo et al., 1992; Irwin and Lee, 2003). Regardless of the energy expenses incurred during breeding, treefrogs should rebuild nutrient supplies as soon as possible in preparation for winter dormancy. Indeed, Kovacs et al. (2007), in a study of the diet of congener Hyla arborea, reported higher feeding intensity in the period prior to dormancy (August-September).
The selective pressure to maintain nutrient levels during the breeding season, and to rebuild nutrient supplies for winter dormancy should be detectable in the form of weight shifts in individuals during the summer months. If true, this would suggest that current monitoring studies for treefrogs should include weight measurements during the summer. We hypothesized a detectable decline in weight during the main part of the breeding period (May – June) for a population of treefrogs in Nodaway County, Missouri. We further hypothesized that this would be followed by a detectable weight gain in the subsequent period (July – August).
Methods
This study took place at Northwest Missouri State University and The Mozingo Outdoor Education Recreation Area (MOERA) in Nodaway County, Missouri during the summers of 2016– 2018 (Lat-Lon: 408 260 2600 N, 948 460 1600 W). Nodaway County receives about 94cm of rainfall each year (U.S. Climate Data). Three ponds and two inlets of neighboring Mozingo Lake were sampled. Ponds and inlets were located an average (6 SD) of 456.4 6 214.66 m apart from each other. Ponds were small with the following approximate areas and depths: Pond A: 300 m2, ~1 m deep; Pond B: 975 m2, ~1m deep; Pond C: 1950 m2, ~1.5 m deep; Inlet D: 6000 m2, ~1.5 m deep; Inlet E: 7480 m2, ~1.5 m deep. Pond A would typically dry in August. Pond B dried up once in August 2018. Inlets D & E also receded significantly in August 2018. The ponds are not stocked with fish, nor periodically inundated with water from the lake, and would presumably have lower predation pressure from fish than inlet sites. At each pond or inlet, within a 50m buffer from the water’s edge, I randomly selected four trees to act as a centering point for four PVC pipes each. This placed the majority of pipes close to the water. The distance of the pipe furthest from the water ranged from 2.72 m to 16.25 m (mean 6 SE: 6.79 6 0.71) thus closely matching the 15 m buffer of highest breeding activity found by Johnson and Semlitsch (2003). Each array of four pipes were arranged such that a 0.6 m pipe was attached to the tree hanging vertically at about 1.5 m height, with the other 3 1-m tall pipes being hammered into the ground standing (ground-secured) upright at these locations: 2 m away from the tree towards the water, 5 m away from the tree towards the water, 2 m away from the tree in the direction opposite the water. Boughton et al. (2000) found that pipes attached to trees were most effective at 0.6 m in length attached at 2 m high and this is the reason for the size difference for ‘‘tree pipes’’.The trees pipes were attached to were Honey Locust (Gleditsia triacanthos), Eastern Red Cedar (Juniperus virginiana), Black Willow (Salix nigra), and Osage Orange (Maclura pomifera). A total of 16 pipes (four arrays) were placed at each pond or inlet. Pipes were 3.8 cm in diameter (Boughton et al., 2000; Pittman et al., 2008). Pipes were checked twice per week during each sampling season which started in early May and ended in late August. Johnson and Semlitsch (2003) found some evidence of breeding (oviposition in artificial pools) as late as mid-August in their study site in south-central Missouri. Downloaded from http://meridian.allenpress.com/tmas/article-pdf/48/2020/17/2877477/i0544-540x-48-2020-17.pdf by guest on 08 December 2025MISSOURI ACADEMY OF SCIENCE: J. D. McGhee 19 If a treefrog was in a pipe, it was gently pushed using a metal rod into a plastic bag, measured for snout-vent length (svl) in mm, weighed to the nearest gram, and sexed based on throat coloration. A new bag was used for each individual captured except when multiple individuals used the same pipe. Treefrogs were marked with visible implant alphanumeric tags (VI Alpha tags, Northwest Marine Technology, Inc, Shaw Island, Washington, USA) under the skin in the tibiofibular region of the leg. Clemas et al. (2009) found that VI Alpha tags were a reliable method for marking anurans. The injection needle for the tag was sterilized with isopropyl alcohol before and after each injection of a tag, the incision site was closed with New Skint (Moberg Pharma North America), and the treefrog was returned to its pipe. I used a contingency table analysis (Zar, 1999) to test the independence of pipe usage by site (Ponds A, B, C, Inlets D, E), pipe location (on tree, 2m past tree, 2m from tree towards pond, 5m from tree towards pond), and year (2016, 2017, 2018). As with Johnson et al. (2008), only unique pipe-frog captures were analyzed since treefrogs would be found in the same pipes in subsequent sampling periods. I used a chi-square test to determine if there was a significant bias in sex ratio across years for each site and I used a one-way ANOVA to test for average sex ratio differences between water bodies. For all statistical tests, a ¼ 0.05, and Bonferroni adjustments were made to account for multiple chi-square tests and subdivided contingency tables.
Results
Over the course of three field seasons, I marked 148 treefrogs in the three ponds and two inlets (62 in 2016, 29 in 2017, 57 in 2018). We identified the sex of 138 individuals (58 females and 80 males) for an overall sex ratio of 0.73 females per male. Mean monthly detection rates (the probability of detecting a treefrog in a PVC pipe) were (6 SE) 0.12 6 0.01 in May, 0.09 6 0.01 in June, 0.14 6 0.01 in July, and 0.15 6 0.01 in August. Daily detection rates averaged (6 SE) 0.12 6 0.01 in 2016, 0.09 6 0.01 in 2017, and 0.16 6 0.01 in 2018. The proportion of pipes that were occupied by a treefrog at least once over the sampling period was 0.53 in 2016, 0.36 in 2017, and 0.61 in 2018. Recaptures were common, with many individuals being found in the same pipe (one male, K38, was found in the same pipe 38 times over a three year period). The combination of categories for contingency table analysis (pipe location x site x year) allowed for a number of zeros to occur in the data set (Feinberg 1970). As a solution to this, I conducted a heterogeneity test to determine if the data could be pooled by year (Zar 1999). The outcome of the Table 1. The number of unique frog detections by pipe location from three ponds and two lake inlets of Mozingo Lake, Nodaway County, MO for May through August 2016–2018. Pipe locations refer to the placement of PVC relative to a randomly selected tree within 50 m of the pond’s edge. Numbers represent the number of times a unique individual frog occurred in a pipe. Recounts of individual frogs only occur if it moved between pipes. Sex ratio refers to the number of females per male for each water body pooled across the three year period. Water Body Pipe Location Pond A Pond B Pond C Inlet D Inlet E 2 m away of tree 10 on tree 2 2mtowardswater 3 5mtowardswater 1 Sex Ratio 0.25 9 6 10 6 0.19 19 6 12 32 10 32 19 6 9 1 29 6 0.92 1.65 0.13 heterogeneity test suggested that this was reasonable (G ¼ 23.40, df ¼ 24, P ¼ 0.4966). The pooled contingency table analysis did detect a difference between pond and inlet use (G ¼9.00, df ¼ 3, P ¼ 0.0267), so I subdivided the contingency table to compare each individual site to all other sites (Bonferroni adjustment of a, ¼ 0.01) and found that this difference was driven by inlet D in which we detected significantly more treefrogs than either the ponds or inlet E (G¼13.61, df¼3, P¼0.0035, Table 1). I also compared each pipe location to all other locations (Bonferroni adjustment of a ¼0.0125), but found that pipe location had no significant effect on use by treefrogs (A: G ¼ 4.43, df ¼ 3, P ¼ 0.2183; B: G¼ 2.00, df ¼3, P¼0.5720; C: G¼10.76, df¼3, P¼0.0131; E: G ¼ 2.06, df ¼ 3, P ¼ 0.5597). The overall sex ratio (female:male) in pond B (0.19) and inlet E (0.14) was significantly male-biased (B: v2 ¼ 11.56, df ¼ 1, P ¼ 0.0006; E: v2 ¼ 9.94, df ¼ 1, P ¼ 0.0016), but no other sites were significantly different from 1:1 (Bonferroni adjustment of a ¼ 0.01; A: v2¼3.60, df¼1, P¼0.0578; C: v2¼0.04, df¼1, P¼ 0.8415; D: v2¼3.69, df¼1, P¼0.0549). I also conducted the chi-square analysis limiting the data to the primary breeding months of May and June (Johnson 2000). Under this constraint, only pond B was male-biased (0.2 female:male; v2¼8.00, df¼ 1, P¼0.0047). Sex ratio in the other water bodies in May and June was not different from 1:1 (Bonferroni adjustment of a ¼ 0.01; A: v2¼2.00, df¼1, P¼0.1573; C: v2¼2.57, df¼1, P¼ 0.1088; D: v2¼1.20, df¼1, P¼0.2733; E: v2¼5.44, df¼1, P ¼0.0196). Mean sex ratio between water bodies varied but did not significantly differ (F2,12 ¼ 0.5799, P ¼ 0.5749).
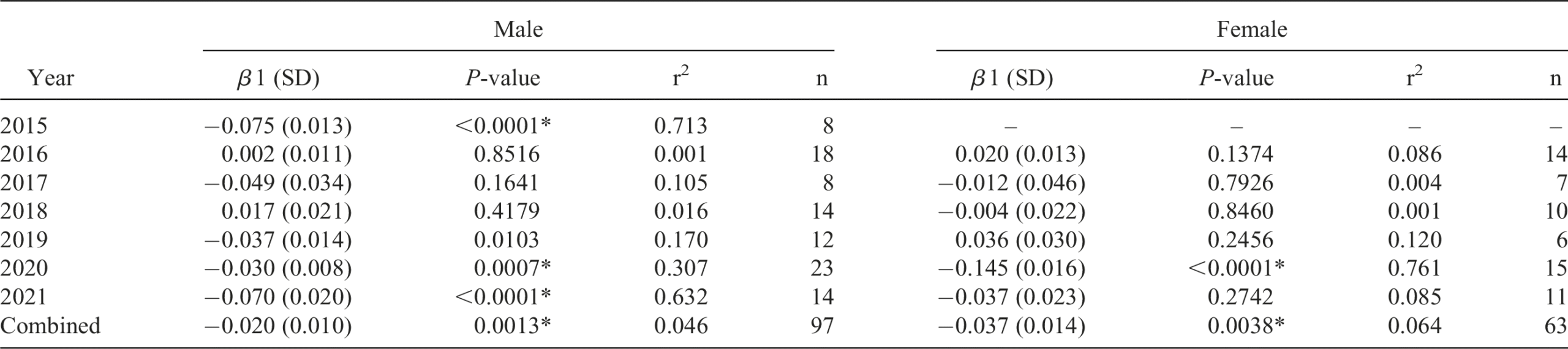
Discussion
Overall patterns of weight change matched our hypotheses for both sexes. We documented weight declines in May-June, followed by weight increases in July-August. Nevertheless, our results varied highly by year and sex. Significant weight loss during May-June appeared to be more commonly true for males than females. This difference between the two sexes likely reflects the heightened energy requirements males undergo by calling (Taigen and Wells, 1985). However, even for males, in some years significant weight loss was not detected. It is possible that male energy loss over this time is usually offset by foraging bouts during the day or during absences from chorusing (Murphy, 1994; Wells et al., 1995; Johnson et al., 2007). As such, measuring weights on a weekly basis would not be precise enough to detect energy losses in most years. Instead, we would be detecting only unusually poor periods where energy losses in the form of weight loss would last for a longer period. This would be true for females as well, but our results suggest that significant weight loss occurs in fewer years during this period than for males. Females during this period may travel to assess multiple breeding ponds and tend to locate at the periphery of a population’s distribution possibly for this purpose or to avoid foraging competition with males who are in higher densities around breeding ponds (Johnson et al. 2007). Despite the greater degree of movement and consequent energy cost this may require, it is likely only for a short period of time, perhaps as short as 48 hours in some populations (Johnson et al. 2007), and so was not detectable by this study (sampling two consecutive days per week).
Later in the summer (July-August), both sexes show significant weight gains in three of seven years, with overlap in years 2016 and 2020. During this period prior to cooler weather, one would expect increased foraging to offset any costs associated with breeding, but also to prepare for winter dormancy by increasing glycogen stores (Irwin and Lee, 2003; Costanzo et al., 1992). This would presumably result in weight gain as late summer progressed. Our inability to detect these weight gains in most years is again likely due to the coarseness of our sampling strategy, such that we were only able to detect major shifts in weight. Large scale changes in weight may only occur in some years, potentially high-quality years in terms of prey availability, offsetting competitive pressures.
One caveat to our analysis is that individual frogs were tracked at varying rates over the summer. Some were captured multiple times, some were captured just once. Those treefrogs that were reweighed more consistently over the course of the summer contributed more data to the analysis. If treefrogs that were likely to use PVC pipes for longer periods (allowing multiple measurements) differed from the larger population, it would bias our results. However, we were unable to find a significant difference between treefrogs captured only once and treefrogs captured multiple times, suggesting that this was not the case. A second caveat is that the conditions of our study meant that treefrogs could join or contribute a weight measurement at any time. This would be problematic if younger, and presumably lighter treefrogs joined later than older and heavier treefrogs because it would falsely imply a weight loss. We did on occasion see older females who had survived the winter start using pipes early, be measured a couple of times, then not be captured again, but this was rare and would be unlikely to bias results heavily. Other bias could include environmental factors such as general weather conditions and a short sampling period that excluded the months of March, April, September, and October.
Our results suggest that there are detectable weight shifts in some years during the summer, presumably reflecting a reproductive season (May – June) and winter preparatory season (July–August). This indicates that the summer period overall (May–August) in northwest Missouri is an ecologically important period for treefrogs, requiring the balancing of multiple goals, such as reproducing and preparing for winter. In addition, there is an inherent variability in annual conditions affecting treefrogs’ ability to meet these goals. We suggest monitoring protocols for populations should include weight measurements, especially during summer.
Acknowledgments I would like to thank B. Lang, S. Olson, A. Athen, A. Slagle, H. Bassich, T. Sutton, R. Dawson, C. Bridgeman, and K. Nowack for their on-going help with field work for this project. Field data were collected under a Missouri scientific collector’s permit (15836, 16715). Frogs were sampled under IUCAC number 200102.
Literature Cited
Biek, R. W., C. Funk, B. A. Maxell, and L. S. Mills. 2002. What is missing in amphibian decline research: insights from ecological sensitivity analysis. Conservation Biology 16: 728–734. Blaustein, A. R., J. M. Romansic, J. M. Kiesecker, and A. C. Hatch. 2003. Ultraviolet radiation, toxic chemicals and amphibian population declines. Diversity and Distributions 9: 123–140. Downloaded from http://meridian.allenpress.com/tmas/article-pdf/48/2020/17/2877477/i0544-540x-48-2020-17.pdf by guest on 08 December 2025MISSOURI ACADEMY OF SCIENCE: J. D. McGhee 21 Boughton, R. J., J. Staiger, and R. Franz. 2000. Use of PVC pipe refugia as a sampling technique for hylid treefrogs. American Midland Naturalist 144: 168–177. Clemas, R. J., J. M. Germano, R. Speare, and P. J. Bishop. 2009. Use of three individual marking methods in Australian frogs (Genus: Litoria) with notes on placement of visible implant alphanumeric tags. New Zealand Natural Sciences 34: 1–7. Feinberg, S. E. 1970. The analysis of multidimensional contingency tables. Ecology 51: 419–433. Harper, E. B., and R. D. Semlitsch. 2007. Density dependence in the terrestrial life history stage of two anurans. Oecologia 153: 879–889. Houlahan, J. E., S. C. Findlay, B. R. Schmidt, A. H. Meyer, and S. L. Kuzmin. 2000. Quantitative evidence for global amphibian population declines. Nature 404: 752–755. Johnson, J. R., and R. D. Semlitsch. 2003. Defining core habitat of local populations of the gray treefrog (Hyla versicolor) based on choice of oviposition site. Oecologia 137: 205–210. Johnson, J. R., J. H. Knouft, and R. D. Semlitsch. 2007. Sex and seasonal differences in the spatial terrestrial distribution of gray treefrog (Hyla versicolor) populations. Biological Conservation 140: 250–258. Johnson, J. R., R. D. Mahan, and R. D. Semlitsch. 2008. Seasonal terrestrial microhabitat use by gray treefrogs (Hyla versicolor) in Missouri oak-hickory forests. Herpetologica 64: 259–269. Johnson, T. R. 2000. The Amphibians and Reptiles of Missouri. Jefferson City, Missouri: Missouri Department of Conservation. Kats, L. B., and R. P. Ferrer. 2003. Alien predators and amphibian declines: review of two decades of science and the transition to conservation. Diversity and Distributions 9: 99–110. Lanoo, M. J., K. Lang, T. Waltz, and G. S. Phillips. 1994. An altered amphibian assemblage: Dickinson county, Iowa, 70 years after Frank Blanchard’s survey. The American Midland Naturalist 131: 311–319. MacKenzie, D. L., J. D. Nichols, G. B. Lachman, S. Droege, J. A. Royle, and C. A. Langtimm. 2002. Estimating site occupancy when detection probabilities are less than one. Ecology 83: 2248–2255. Marsh, D. M., and P. C. Trenham. 2001. Metapopulation dynamics and amphibian conservation. Conservation Biology 15: 40–49. Pittman,S.E.,A.L. Jendrek,S. J.Price, andM.E.Dorcas. 2008. Habitat selection and site fidelity of Cope’s gray treefrog (Hyla chrysoscelis) at the aquatic-terrestrial ecotone. Journal of Herpetology 42: 378–385. Rothermel, B. B., S. C. Walls, J. C. Mitchell, C. K. Dodd, Jr., L. K. Irwins, D. E. Green, V. M. Vazquez, J. W. Petranka, and D. J. Stevenson. 2008. Widespread occurrence of the amphibian chytrid fungus Batrachochytrium dendrobatidis in the southeastern USA. Diseases of Aquatic Organisms 82: 3–18. Semlitsch, R. D. 2000. Principles of management of aquatic breeding amphibians. Journal of Wildlife Management 64: 615–631. Smith, G. R., J. E. Rettig, G. G. Mittelbach, J. L. Valiulus, and S. R. Schaack. 1999. The effects of fish on assemblages of amphibians in ponds: A field experiment. Freshwater Biology 41: 829–837. Storfer, A. 2003. Amphibian declines: future directions. Diversity and Distributions 9: 151–163. U.S. Climate Data. 2020. Climate Maryville– Missouri. https:// www.usclimatedata.com/climate/maryville/missouri/unit ed-states/usmo0565. Accessed 24 Oct 2020. Zacharow, M., W. J. Barichivich, C. K. Dodd, Jr. 2003. Using ground-placed PVC pipes to monitor hylid treefrogs: Capture biases. Southeastern Naturalist 2: 575–590. Zar, J. H. 1999. Biostatistical Analysis, 4th Edition. Prentice Hall, Upper Saddle River, NJ.
